São Muitos Anos A Virar Frangos
Week Five In The LabLooking back on my time in the lab at FFU Lisboa, there is one major learning point that summarizes my experience. Prior to this rotation, I had a vague idea of the research process and the steps that needed to be taken to get a medication on the market. When you are learning about the different tests that must be performed on a new entity, they all seem so simple on paper. However, what I discovered is the volume of laboratory procedures that are completed to be able to characterize a medication is rather large. This is primarily due to the fact that each test is not run once and done. The same test is completed multiple times in order to ensure reproducible results, and then it is also performed again under slightly different conditions, and again, and again...to optimize the results. Then the same process occurs for the next measurable parameter until the drug's characteristics are fully defined. All of this has to be executed as part of the preclinical studies, before the medication is ever even given to animal or human research subjects.
With respect to the individuals in the laboratory that dedicate their career to this process, they all have certain attributes that allow them to be successful researchers. They are extremely detail oriented, constantly checking and double checking that every setting for every experiment is perfect and also documenting the information in their laboratory book religiously. They are incredibly patient; a large part of performing research is waiting for the test to run its course often taking several hours or days. They are dedicated, spending long hours in the lab day after day doing somewhat monotonous tasks is not an easy job. They are well educated; in addition to their college education course, which may include masters or doctorate degrees, these individuals are constantly attending conferences and reading journal articles to stay up to date on the latest developments in their field and the research community in general. I could probably go on, but I think you get the idea that I admire these individuals and the work they do very much. Just as a closing note, I would like to thank Professor Helena Marques for her assistance in arranging this amazing rotation opportunity and agreeing to act as my preceptor. Additionally, a big thank you to everyone at the Faculdade de Farmácia da Universidade de Lisboa (Faculty of Pharmacy of the University of Lisbon) for explaining to me their many projects, methods, and equipment while allowing me to feel like a real member of their research team.
31 Comments
This week I had the opportunity to travel to Lumiar, the second campus of iMed that houses additional laboratories. I was able to observe the research team studying nanoparticles on the project Cutaneous leishmaniasis: an innovative idea to explore the topical treatment of skin lesions. For some information about Leishmaniasis you can read a brief description below and also navigate the websites for the Centers for Disease Control and Prevention (CDC) or the World Health Organization (WHO). The two PDF files are recent publications on this topic from the iMed research group. Leishmaniasis
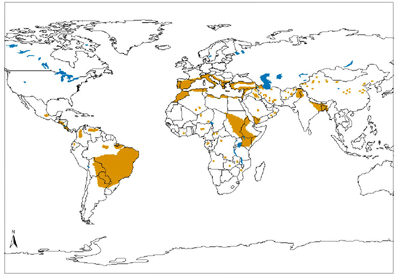 Distribution of Visceral Leishmaniasis in the world; map available from the World Health Organization Distribution of Visceral Leishmaniasis in the world; map available from the World Health Organization Leishmaniasis is a disease that is caused by a parasite and transferred to humans by the bite of an infected sandflies. There are three forms of leishmaniasis: cutaneous, visceral, and mucocutaneous. The most common form of leishmaniasis is cutaneous which causes ulcers on the body that can lead to disfigurement or scarring. Visceral leishmaniasis is the most severe form, fatal if untreated, affecting the most vital organs of the body causing enlargement of the liver and spleen. Destruction of the membranes in the nose, mouth, and throat is caused by the mucocutaneous form of leishmaniasis. Since many images of leishmaniasis infections are not for the feint of heart, I am not going to spare you by not including a picture here. However, there are many available on the websites below or by performing a Google Images search. Risk factors for leishmaniasis include living in a rural area or traveling to an endemic area, poverty (due to living conditions), malnutrition, and urbanization (developing previously forested areas). Additionally, individuals co-infected with HIV have higher rates of clinically significant disease and resultant morbidity/mortality. Diagnosis of leishmaniasis occurs by performing specific laboratory testing, such as examining tissue samples with a microscope, for patients with clinical suspicion of the disease. In the United States, treatment of a patient infected with leishmaniasis is worth consultation with the Centers for Disease Control. Medications used to treat leishmaniasis such as meglumine antimoniate, liposomal amphotericin B and conventional amphotericin B are on the World Health Organization Model National Essential Drug List (a list of medications that are a priority for the health needs of a country and are intended to be available within the health system at all times at an affordable price). Sources/Additional Available Reading
 Torre dos Clérigos Torre dos Clérigos Friday night we kicked off the weekend with a tri-birthday celebration. Since we did not want to miss the festivities, that meant Irma and I took the bus to Porto at 7 am on Saturday morning to arrive in the north by 10:30 am. Although we were rather exhausted by the time we arrived, we wasted no time in starting to explore since we had just two days to experience all of Porto. The second largest city in Portugal, Porto has existed since the medieval time period demonstrated by the buildings of the historic center named as a World Heritage Site. Irma has several friends from the city and has visited many times before, so she was familiar with what to see and what to do. The first point of interest was the Torre dos Clérigos (Clérigos Tower). This baroque-style tower has become a symbol synonymous with the city of Porto itself. It pierces the skyline with its unique silhouette, visible from almost viewpoint. The tower itself was designed by Nicolau Nasoni, an Italian architect. His body was buried somewhere under the church attached to the infamous tower, but the exact location is unknown. While we were there, we did take advantage of two different free walking tours offered of the city. The first was on Saturday afternoon and focused on the newer, more modern part of the city. The second was on Sunday morning and covered the old part of the city that had previously been encapsulated by a large city wall for protection. Both of the tours had a special flavor with interesting stories and myths told about the various monuments and traditions of Porto. For instance, the people of Porto are sometimes referred to as tripeiros. This literally translates to mean guts. At first thought to be an unappealing and derogatory term, it turns out there is a rich history behind this nickname. Porto or Oporto = the port. It is obvious why the city has this name when you consider it is situated on the mouth of the Rio Douro (Douro River) opening into the Atlantic Ocean. Therefore, this was a major site for exploration and trading. When the ships used to leave the port to go on long journeys, all the people would give the sailors meat for the journey keeping only the guts of the animals to eat for themselves. Calling the people tripeiros is in reference to this unselfish display of generosity toward one another. To this day, guts are still considered a delicacy in Porto. (Additional pictures from Porto with descriptions have been added to the Photo Gallery)
Therefore, the monument is not just a religious symbol but also captivates the meaning of peace among men from its history intwined with a time of war. Building the monument required about 40 thousand tons of concrete. The figure of Christ is erected more than 100 meters from the ground (3 meters for the base, 82 meters for the pedestal, and 28 meters for the statue of Christ) and was carved by hand. It is designed to appear as Christ embracing the city of Lisbon and the whole country in His arms. Spray Drying
The final powdered product is most often collected in a cyclone where the flow of air separates the unwanted particles from the desired particle size. Particles that are too small adhere to the wall of the cyclone, particles that are too large are filtered into a waste container, and the desired particles are deposited into the collection container at the end of the system. An advantage to spray drying over other techniques (such as oven drying or freeze drying) is the relative uniformity and sphericity of particles produced. Adjusting the settings on the spray dryer allows for control of particle properties such as density, size (in micrometers or nanometers), and moisture content. Settings that can be altered depend on the specific spray dryer but may include the concentration of the liquid, feed rate of the liquid, aspirator flow rate or air flow rate, and inlet temperature or temperature of the air running through the drying chamber. The temperature of the air at the point in which the particles pass into the collection cyclone is known as the outlet temperature, which cannot be controlled directly but is a dependent variable of the other previously mentioned settings. The video below gives a brief demonstration of one type of spray dryer, which is similar to the one I used, so you can see the concepts I have described.
I haven't really posted much this week other than catching you all up on what I was doing in the laboratory. Mostly, that was because nothing outrageously exciting has been going on that I considered to be "out of the normal." Then, yesterday, I saw my cousin posted a photo of a new home decoration she bought that read "Enjoy the little things in life, for someday you will realize they were the big things." And all of the sudden I gained a new perspective on this past week and although it may have seemed slightly insignificant compared to previous posts, it really has been far from that. After all, I'm living in Portugal!  I had the opportunity to meet up with some of the SEP students that I met in Figueira da Foz to explore Belém and see the Tower of Belém (unfortunately we got there too late we couldn't go inside; fortunately that meant I was able to take this wonderful sunset picture). Although I didn't get a picture of it (yet), we used the old-fashioned classic yellow tram to get there. If you're familiar with the inclines in Pittsburgh, it can be compared very closely to that. The tram could probably qualify as functional antique, as the rickety ride at some times felt as if it may fall apart similar to the Wildcat roller-coaster at Hershey Park. Of course once we arrived at our destination we also stopped at the bakery Pastéis de Belém to sample their version of pastel da nata, which is rumored to be the absolutely best version available. Afterward, I was able to show the girls around some of my favorite parts of Lisbon as I had done with the Germans. Seriously, I should start charging money for being a Lisboa tour-guide. I know I keep talking about all the viewpoints, but I have to share my latest photo from a completely different angle of the city. This was taken from the top of the Avenida da Liberdade (Avenue of Liberty) which is the main mile-long avenue of the city running from Restauradores to Marquês de Pombal Square. This important square was named after the man who was the major driving force of Lisbon's reconstruction after an earthquake in 1755 that immensely impacted the city. You can somewhat seen the statue of him that is erected in the center of the roundabout of the square (tall white pillar in the center of the photo). Although further down the avenue there are fancy designer stores and ritzy hotels, at the very top you will notice it is a lush green landscape with pathways for walking. Behind me, therefore not in the picture, was also a gigantic Portuguese flag flying in the wind. All in all, there is just a large amount of respect for the history of the city and country present at this very spot.
And finally, to wrap up this week, today I visited the much anticipated Oceanário de Lisboa (Lisbon oceanarium). It certainly did not disappoint. The main exhibit was laid out in the shape of a square. Each corner tank represented the four main ocean habitats of the world (North Atlantic, Antarctic, Temperate Pacific, and Tropical Indian). The center exhibit was called the Global Ocean and took into account how all of the oceans of the world are connected through continuous flowing water. Therefore, this exhibit was the largest of all and accentuated how, in theory, ocean animals could venture anywhere on Earth if it were not for physiological/anatomical limitations. The entire building appeared to be one gigantic ocean with all sizes, shapes, and colors of life swimming about. You can see a few of my favorite photos below. Hope you enjoy!
Cyclodextrins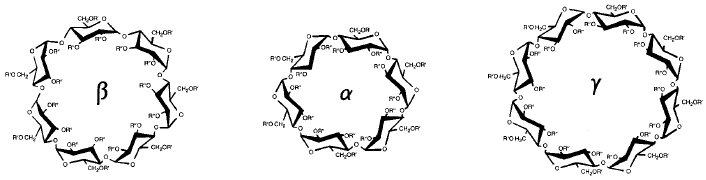 Image available at: http://www.sigmaaldrich.com/technical-documents/articles/biofiles/cyclodextrins.html Image available at: http://www.sigmaaldrich.com/technical-documents/articles/biofiles/cyclodextrins.html Some drugs are simply not very compatible with the human body. The chemical characteristics of these drugs with therapeutic potential are not ideal for actually delivering the substance to the target area. One way in which this is overcome is through the utilization of cyclodextrins. The research project I worked on this week focused on the use of cyclodextrins for drug delivery. As the human body is largely made up of water, cyclodextrins allow substances that are hydrophobic (having low water solubility) to be encapsulated in a carrier that is hydrophilic (with high water solubility) and is capable of dissolving. Cyclodextrins are made up of several simple sugars, monosaccharides, linked to form a cyclical polymer or oligosaccharide. The central cavity is hydrophobic (attracted to lipids or fats) and the outer surface is hydrophilic, as previously mentioned. Although cyclodextrins occur naturally, most of those used for drug delivery are chemically engineered with minor substitutions to increase their water solubility. Several different sizes of cyclodextrins are produced, with the specific cyclodextrin used for drug delivery partially dependent on the size of the drug that is being complexed within the center for administration. For instance, β-cyclodextrin, composed of 7 glucose units, is commonly used as a vehicle for hormones or vitamins due to its appropriate size parameters for these molecules. Since cyclodextrins are generated from starches linked through enzymatic reactions, they are non-toxic to humans and fairly stable within the body. The inclusion of a drug in the core of a cyclodextrin is predominantly a reversible process through dilution of the cyclodextrin-drug complex in aqueous media (water). The cyclodextrin is then subsequently removed from the body either through digestion by bacteria in the gastrointestinal tract or it is excreted in the urine, primarily unchanged. In addition to improving the water solubility of a drug, cyclodextrins may also be employed for stability or palatability reasons. Not only are cyclodextrins being utilized in the pharmaceutical industry, but they also are being used for other purposes such as in food and cosmetics. Cyclodextrins have achieved status as a Generally Recognized As Safe (GRAS) substance according to the Food and Drug Administration (FDA). Sources/Additional Available Reading
Students MeetingThis week I also had the opportunity to attend the 7th iMed.ULisboa Postgraduate Students Meeting which took place at the faculty of University of Lisbon. It was a two-day conference on July 15th and 16th organized by the PhD student commission. On the first day, there were 15-20 minute presentations given throughout the day by various PhD students on the work they are performing in their specific department. Two poster sessions were held during the breaks between sessions for students that may not have had the chance to give a formal presentation to highlight and discuss their work. The second day was geared toward industry and regulatory topics with guest speakers sharing information about the opportunities available at pharmaceutical companies and the primary research topics currently under investigation. All of the presentations were given in English so it was easy for me to follow along and understand. You can see the flyer advertising the event as well as the schedule of speakers and their presentation titles below. During the first week of rotation, I assisted one of the graduate fellows, João, on the project Powdered medicinal milks for oral delivery of drugs in paediatrics (MedMilk). As a part of my first week, I learned about the confidentiality of research in respect to non-human subjects. Previously, my interactions with confidentiality of information have primarily been focused upon patients or research involving patient information (i.e. chart reviews, health questionnaires). Upon making my first attempt at writing this blog post, I simply did as I normally do for this blog and detailed what exactly I had been doing in the laboratory for this project. Then it came to my attention that this could potentially be a breach in confidentiality of the project’s integrity. Although we have no human subjects whose names, birth dates, past medical history, etc. must be kept known to only those directly involved in patient care, there is still a standard of confidentiality applied. This is partially due to the fact that the world of research, drug design and development, is highly competitive. If information relating a group’s project were to be replicated and published by a different research team, the credit for this work could be assigned elsewhere from the original group. Additionally, another motivating factor for confidentiality pertains to the process of obtaining a patent. In order for a patent to be granted, there are several requirements such as novelty of idea, an innovative step, capable of being useful, considered patentable under law, and reproducible. Patents provide incentive to society for inventing by providing exclusivity rights which can translate into monetary gain. To be granted a patent, it cannot be information that is widely available. If there were to be a leak of research information or something published publicly that compromises the integrity of the project it could result in the loss of patent rights. Without the patent, there is no longer profit incentive for a researcher or pharmaceutical company to continue the project. Therefore, even though what I have been working on is intriguing and exciting (to me at least), I will not be able to share the intimate details of my daily lab work. Instead of talking about the details of the projects I will be working on I am going to focus the rotation portion of this blog on different assays and analytical methods that are used in drug design and development laboratories. These tests are necessary to characterize the substances that make up medications. By understanding the properties of the “medication ingredients” we can have a better idea of how the medication will work in the body. In pharmacy terms we usually refer to pharmacokinetics as what the body does to a drug. This describes the absorption, distribution, metabolism, and excretion of a drug upon entering the body whether it is by the oral, transdermal, parenteral, or any other route of administration. After that, pharmacodynamics comes into play, also known as what the drug does to the body. This term is related to the desired effect or outcome of taking the medication, for example, how Tylenol (acetaminophen) works on the body to relieve pain and reduce fever. All of the processes related to both pharmacokinetics and pharmacodynamics can be influenced by a multitude of variables such as genetics, age, organ function, and body composition. Therefore, the more we know about how a drug behaves under various circumstances, the better we can predict how we think it will act in patients. Contact Angle and Wetting Properties When a liquid comes into contact with a solid substance, the interaction between the two results from a contact angle. This term helps indicate the wettability of a substance, or the ability of a liquid to spread out on and adhere to a solid substance. For example, a powder should be wettable to easily dissolve into a solution. In order for a substance to be considered wettable, the contact angle must be <90°. An angle of >90° indicates that the substance is not wettable. For a powder that is non-wettable to dissolve into solution a wetting agent, or another substance that is added to the powder, would be used to change the properties and allow dissolution. Measuring the contact angle of a solid with several liquids can reveal the surface free energy of the solid, which is similar to the surface tension a liquid displays. The classic example of surface tension of a liquid is placing drops of water on top of a penny until a dome of water forms that exceeds the edges of the penny due to intramolecular attraction. The same idea can be applied to the surface of a solid through the surface free energy to indicate the characteristic intermolecular interactions occurring. A contact angle can be measured several ways, one of which is through a dynamic contact angle method. The solid substance is dipped into the liquid substance to generate the advancing contact angle, on the way into the liquid, and the receding contact angle, on the way out of the liquid. With the Wilhelmy plate method, the force required to insert the solid, placed on a vertically suspended plate, into the liquid surface is used to calculate the contact angle. In order to perform this test, advanced equipment, such as a force tensiometer, is utilized for precision and accuracy in measurements. You can see a picture of the one we have available in our lab to the left. Behind the glass doors, the clip in the middle holds the sample of the solid being tested with the liquid in the glass cup below. The pedestal on which the liquid is set lowers up and down to insert and withdrawal the solid sample into/from the liquid using the remote control pictured on the right of the machine. I was very pleased the other day when João remarked that the contact angle samples I had prepared were testing very well and followed the pattern he was expecting. He even told me I could stay in Portugal to continue to help as long as I would like! I am tempted to take him up on this offer… Additional available reading: Contact Angle and Wetting Properties Above is the group picture taken at the "going away" picnic from SEP weekend in Figueira da Foz. As you can probably tell from the photo, it was a fun and dynamic group of students. We had a blast getting to know each other while relaxing at the beach. Many new friends were made as we shared about our different countries and experiences we have had so far in the various cities of Portugal.
I also learned some new information about pharmacists in Portugal. Since I spent a majority of my time with the Portuguese students, we had several discussions about their perspective on the career. Pharmacy is a well-respected profession in Portugal with patients valuing and trusting the advice of their pharmacist concerning their medications. Student pharmacists complete three years of undergraduate study then two years of pharmacy classes with six months spent doing internships full-time. The community aspect of pharmacy is the most common career path, as is reflected in the mandatory 4 month internship that is completed in their final year of school compared to the 2 months spent in hospital pharmacy. Coimbra is the only university that has industry as an option for fulfilling the required internship experiences, for other universities industry is considered to be an extracurricular internship. Therefore, a pharmacist's primary function is similar to that of a community pharmacist in the United States. They dispense medications in an outpatient setting and are available for counseling and medication recommendations. Hospital pharmacy is more difficult to pursue as there are not as many jobs available in this field. There is not a distinction between staff and clinical pharmacists in the hospital, so these individuals are expected to do both to some extent. A typical pharmacist's salary in Portugal is about 800-1,000 Euro per month or approximately 12,000 Euro per year which is about $13,000 USD at the current exchange rate. Although this is considered an average/above average salary in Portugal, the students were shocked (to say the least) when I informed them that pharmacists in the USA make somewhere around $80,000-$120,000. Overall, I gained the sense that the motivation to become a pharmacist in Portugal is similar to that in the USA, a combination of the desire to help patients while being a respected professional by those in the community.  Regina and I reunited after meeting in Turkey a year ago Regina and I reunited after meeting in Turkey a year ago Thursday evening, I took the bus from Lisbon to Coimbra to visit Regina, the other Portuguese girl that I met in Turkey last summer. She studies at the pharmacy faculty Universidade de Coimbra (University of Coimbra) which is actually a World Heritage Site. Coimbra is basically the definition of a U.S. college town, they refer to it as "the city of students" and "the city of doctors" (because it used to be the only medical school in the country). In simplistic terms, the closest analogy for me would me like how State College exists largely because Penn State University is there. Since it is during the summer, many of the students are now at home and the city was less busy than it would be during the semester. We visited the university and toured several of the preserved rooms from when the school was originally built. Regina explained to me many of the school's myths and traditions as we wandered around the campus. For instance, they have a school uniform that includes a long black cape worn on the shoulders. If you're thinking that sounds like Harry Potter, you might be right! Turns out J.K. Rowling lived in the Portuguese city of Porto and the uniforms the students of Hogwarts wear are rumored to be inspired by the Portuguese university uniforms she saw during her time there. Visiting the library, in particular, was breathtaking with its beauty of intricate design and the shear volume of old books collected there. Interestingly enough, it houses many bats that do not cause problems but rather they eat any bugs/moths that enter the library helping to rid of pests that may damage the works of literature. Photos were not allowed inside many of the rooms we visited, so feel free to click on the link above to gain an idea of what was seen. I also ate another traditional Portuguese meal, Francesinha, which I was skeptical about at first but it turned out to be delicious, of course. The rest of my time in Coimbra we spent walking around the various streets with Regina just talking and catching up. We were able to see the oldest pharmacy in Coimbra, which is still in operation, and is also most likely one of the oldest in all of Portugal. Afterwards, we packed up our things and met several students from Regina's faculty to take the train to Figueira da Foz for the Student Exchange Program (SEP) Weekend! Pharmacy students from many countries including Serbia, Poland, Spain, Hungary, Turkey, and even the United States (one from California, the other from Indiana) are here to spend the weekend socializing at the beach.
|
AuthorCheck out the About Me tab for a brief self-biography of Valerie Nolt. Archives
August 2015
Categories"Travel makes one modest. You see what a tiny place you occupy in the world."
- Gustave Flaubert |
||||||||||||||||||||||||||||||||||||||






























 RSS Feed
RSS Feed
